Table of contents
- Key findings
- Summary
- Results
- All malignant neoplasms (tumours): number of newly diagnosed cases, 2011
- All malignant neoplasms: incidence rates by sex and age group, 2011
- All malignant neoplasms: frequency distribution by sex and age group, 2011
- All malignant neoplasms: standardised registration by region, 2011
- Number of registrations by major sites, 2011
- Cancer registrations, 1971 to 2010
- Cancer registrations: interpretation and data quality
- Users and uses of cancer statistics
- Policy context
- Additional information
- References
- Acknowledgements
- Background notes
- Methodology
1. Key findings
The three most common cancers for men in 2011 remained prostate (25.6%), lung (13.8%) and colorectal (13.6%)
The three most common cancers for women in 2011 remained breast (30.7%), lung (11.6%) and colorectal (11.2%)
Rates of cancer in 2011 among 35–44 year old females were more than double the rates for males in the same age group
In the older age groups, rates of cancer in 2011 were higher in males than in females (37% higher in those aged 65–69, and 63% higher in those aged 85 and over)
Cancer incidence was higher than expected for both sexes in the north of England, and for females in the East Midlands and South West regions
2. Summary
This publication presents data for England on those patients who were diagnosed with cancer during 2011 and whose registrations were received at the Office for National Statistics (ONS) by the end of February 2013. This is the first time that ONS has published 2011 statistics on cancer incidence in England.
Cancer registrations are submitted to ONS by the eight regional cancer registries in England. These data are then loaded onto a database and validated. The extensive checks include the compatibility of the cancer site and the associated histology, and are closely based on those promoted by the International Agency for Research on Cancer (IARC, Parkin, Chen, Ferlay et al, 1994). Once all the expected records for any one incidence year have been received and validated, detailed tables are published (see ‘Results’ section below) on the numbers and rates of all types of cancer by age and sex, and by region of residence.
Back to table of contents3. Results
The three most common cancers, 2011
In the International Classification of Diseases, Tenth Revision (ICD-10), there are 88 three-digit site codes relating to malignant neoplasms (tumours): of these, four relate to males only and eight to females only. For both males and females three cancer sites (different ones for each sex) constituted just over half of the total registrations in 2011. The most common cancer site for males was prostate, and for females breast. The second and third most common cancers for both males and females were lung and colorectal cancer. The number of registrations for each of these cancer sites is shown in Table A.
Table A: The three most common cancers, England, 2011
| ICD-10 code | Site description | Number of registrations | % of total malignancies |
| Males | |||
| C61 | Prostate | 35,567 | 25.6 |
| C34 | Lung | 19,173 | 13.8 |
| C18-C20 | Colorectal | 18,971 | 13.6 |
| Total | 73,711 | 53.0 | |
| All malignancies1 | 139,120 | 100.0 | |
| Females | |||
| C50 | Breast | 41,523 | 30.7 |
| C34 | Lung | 15,675 | 11.6 |
| C18-C20 | Colorectal | 15,073 | 11.2 |
| Total | 72,271 | 53.5 | |
| All malignancies1 | 135,113 | 100.0 | |
| Source: Office for National Statistics | |||
| Notes: | |||
| 1. Excluding non-melanoma skin cancer (ICD-10 C44) | |||
Download this table Table A: The three most common cancers, England, 2011
.xls (55.3 kB)Breast cancer accounted for just under one-in-three newly diagnosed cases of cancer in females. The main risk factor for breast cancer, after gender, is age: 80% of all new cases of breast cancer diagnosed among women in 2011 were among those aged 50 and over (see Table 1 of the reference tables associated with this publication (1.19 Mb Excel sheet)). It is estimated that about 27% of cases of female breast cancer in the UK are linked to lifestyle and environmental factors, such as alcohol consumption, obesity, lack of physical activity, and hormonal and reproductive factors (Parkin, Boyd and Walker, 2011).
Prostate cancer accounted for just over one-in-four newly diagnosed cases of cancer in males in 2011. Prostate cancer incidence has increased greatly since the early part of the 1990s, largely reflecting improvements in diagnosis from increased use of Prostate-Specific Antigen (PSA) testing (Quinn, Babb, Brock et al, 2001). Between 2002 and 2011 the age-standardised rate increased by 10% ( Table 9 (1.19 Mb Excel sheet)). The strongest risk factor for prostate cancer is age: 89% of all new cases in 2011 were in those aged 60 and over (Table 1 (1.19 Mb Excel sheet)).
Lung cancer was the second most common cancer among both men and women in 2011, accounting for 14% and 12% of all newly diagnosed cases respectively. Since 2002 the age-standardised incidence rate has decreased by 11% among males but has increased by 15% among females (Table 9 (1.19 Mb Excel sheet)). The majority of lung cancer cases occur as a result of tobacco smoking, with around one in five cases in the UK being attributable to diet and occupational exposures (Parkin, Boyd and Walker, 2011). While men remain more likel to smoke than women, the gap has narrowed: whereas 51% of men and 41% of women smoked in 1974, 21% of men and 19% of women smoked in 2011 (Office for National Statistics, 2013).
Colorectal (or bowel) cancer was the third most common cancer in both males and females in 2011. The age-standardised incidence rate for colorectal cancer has remained fairly stable for both sexes over the past ten years (Table 9 (1.19 Mb Excel sheet)). As with many cancers, the biggest risk factors for bowel cancer are age and family history (Cancer Research UK, 2012). In 2011, 71% of newly diagnosed cases in men, and 74% of those in women, were among those aged 65 and older (Table 1 (1.19 Mb Excel sheet)).
Full details of registrations of newly diagnosed cases of cancer by site, sex and age group are presented in Table 1 (1.19 Mb Excel sheet) of the reference tables associated with this publication.
Back to table of contents4. All malignant neoplasms (tumours): number of newly diagnosed cases, 2011
Registrations of non-malignant skin cancer (nmsc, ICD-10 C44) are excluded from the figures in the commentary that follows, as experts have advised that it is greatly under-registered.
There were 139,120 new cases of cancer registered for males and 135,113 new cases registered for females in England in 2011. This represents an age-standardised incidence rate of 423 new cases per 100,000 men and 372 new cases per 100,000 women. Since 2002, the age-standardised incidence rate has increased by 3% for males and 8% for females (see Tables 8 and 9 of the reference tables associated with this publication (1.19 Mb Excel sheet)). Much of this increase is due to people living longer: as age increases so does the incidence of cancer (see below).
Back to table of contents5. All malignant neoplasms: incidence rates by sex and age group, 2011
Cancer is predominantly a disease of the elderly. The overall crude rates of cancer registrations (C00–C97 excluding non-melanoma skin cancers (nmsc)), 532 new cases per 100,000 population for males and 501 new cases per 100,000 population for females, conceal wide differences between the sexes and across the age groups, as illustrated in Figure A. Following the small decrease in rates after early childhood, rates increased continuously with age for both males and females. From the 25 to 29 age group up to the 55 to 59 age group, rates of cancer were higher in females than in males, and in the 35 to 39 and 40 to 44 age groups, the rate in females was more than double that for males. Much of this difference is due to the high incidence of breast cancer among women in these age groups: in 2011 breast cancer account for 44% of all malignant cancers (excluding nmsc) among women aged 25-59. Rates of cancer were higher in males than females from the 60 to 64 age group onwards: the difference between the two sexes ranged from 37% in the 65 to 69 age group to 63% in the 85 and over age group.
Figure A: All malignant neoplasms: incidence rates by sex and age group, England, 2011
Source: Office for National Statistics
Notes:
- Excluding non-melanoma skin cancer (ICD-10 C44)
Download this chart Figure A: All malignant neoplasms: incidence rates by sex and age group, England, 2011
Image .csv .xlsThe numbers on which Figure A is based are presented in Table 2 (1.19 Mb Excel sheet) of the reference tables associated with this publication.
Back to table of contents6. All malignant neoplasms: frequency distribution by sex and age group, 2011
Figure B shows the frequency distribution by sex and age group for cancer registrations in 2011. Excluding non-melanoma skin cancers (nmsc), only 1,173 (0.4%) of all malignancies occurred in children aged under 15 years, with the most common of these being leukaemias (ICD-10 C91–C95, 336 malignancies, 29% of all malignancies in children aged under 15). From the 25 to 29 years age group to the 55 to 59 years age group, the higher percentage of cancers in females than males was mainly due to the incidence of cancers of the breast (ICD-10 C50) and cervix (ICD-10 C53). Cancers in those aged under 45 years accounted for 5% of the total for males and 9% for females.
There was a clear peak in the age group frequency distribution for males, with the greatest percentage of cancer registrations being recorded for males aged 70 to 74 years. For females, there was no such clear peak: the greatest percentage of registrations were recorded for ages 60 to 64 years up to 85 years and over.
Figure B: All malignant neoplasms: frequency distribution by sex and age group, England, 2011
Source: Office for National Statistics
Notes:
- Excluding non-melanoma skin cancer (ICD-10 C44)
Download this chart Figure B: All malignant neoplasms: frequency distribution by sex and age group, England, 2011
Image .csv .xlsThe numbers on which Figure B is based are presented in Table 1 (1.19 Mb Excel sheet) of the reference tables associated with this publication.
Back to table of contents7. All malignant neoplasms: standardised registration by region, 2011
Standardised registration ratios (SRRs) allow regions to be compared, with the registration rates for England being taken as standards. If a regional cancer incidence rate is high compared with the national rate, then the observed incidence for that region will be higher than the expected incidence and the SSR will be greater than 100. If a regional cancer incidence rate is lower than the national rate, then the SSR will be lower than 100. However, these SRRs should be interpreted with caution because it is difficult to separate the effect of variation in levels of ascertainment of cases by the cancer registries from genuine geographical differences in incidence. However, it is recognised that there are generally higher levels of deprivation in the north of England than in the south (Communities and Local Government, 2011) and increased incidence rates of many cancers has been associated with higher levels of deprivation (National Cancer Intelligence Network, 2008, Shack, Jordon, Thomson et al, 2008).
Figure C shows that actual cancer incidence was higher than the expected incidence in the north of England (North East, North West, and Yorkshire and The Humber regions) compared with the England baseline. It was also higher for females only in the East Midlands and South West regions.
Figure C: All malignant neoplasms: standardised registration ratios by region, England, 2011
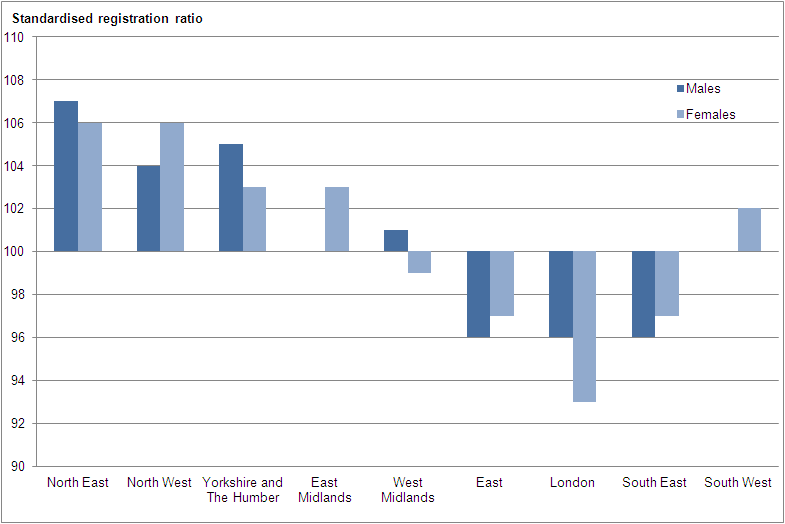
Source: Office for National Statistics
Notes:
- Excluding non-melanoma skin cancer (ICD-10 C44)
Download this image Figure C: All malignant neoplasms: standardised registration ratios by region, England, 2011
.png (16.3 kB)- Excluding non-melanoma skin cancer (ICD-10 C44)
The numbers on which Figure C is based are presented in Table 5 (1.19 Mb Excel sheet) of the reference tables associated with this publication.
Back to table of contents8. Number of registrations by major sites, 2011
Figure D shows the number of registrations for 21 major sites for males and females in 2011. These registrations represented 90 and 89% of the total number of registrations for males and females respectively.
Figure D: Number of registrations by major sites, England, 2011
Source: Office for National Statistics
Notes:
- Excluding non-melanoma skin cancer (ICD-10 C44)
Download this chart Figure D: Number of registrations by major sites, England, 2011
Image .csv .xlsSince 2002, there have been large increases in incidence rates of oral, uterine and kidney cancers, and malignant melanoma of the skin (Table 9 (1.19 Mb Excel sheet)). These cancers are strongly linked to lifestyle choices, such as smoking, alcohol consumption, diet, and overweight and obesity (Parkin, Boyd and Walker, 2011). Malignant melanoma incidence rates have increased by 56% among males and 38% among females. These increases are considered to be due to changes in exposure to solar UV rays as a result of altered patterns of behaviour in recent decades, such as choice of clothing and recreational sunbathing (Parkin, Mesher and Sasieni, 2011).
The numbers on which Figure D is based are presented in Table 1 (1.19 Mb Excel sheet) of the reference tables associated with this publication.
Back to table of contents9. Cancer registrations, 1971 to 2010
Registration of cases of cancer is a dynamic process in the sense that the data files, both in the cancer registries and at ONS, are always open. Cancer records may be amended: for example the site code would be modified should later and more accurate information become available. The date of death is added to the record for those cases registered when the person was alive. Although relatively unusual, records may be cancelled. Also, complete new ‘late’ registrations may be made after either the cancer registry or ONS, or both, have published what were thought at the time to be virtually complete results for a particular year.
Refreshing the database means that current numbers of registrations are generally greater than the published figures. The balance between ensuring completeness of the data but publishing tables in a timely manner is necessarily a compromise. The gap between the data year and production of tables has varied considerably over time, which results in varying proportions of additional cancer registrations held on the ONS database in comparison with the numbers published in the corresponding annual report (MB1 series), as shown in Figure E. Over the 40-year period reported, differences between published and current numbers of registrations for each year average less than 5%.
Figure E: Number of registrations published in the annual report (MB1 series) and currently on the National Cancer Registry database, England, 1971–2010
Source: Office for National Statistics
Notes:
- Figures for 1971 to 1989 have been extracted from the 'frozen' historical data set published in 2012, and are therefore the same as those published in this series last year
- The published figures for 1971 to 1978 correspond to all malignancies, not all registrations
- Figures at February 2013
Download this chart Figure E: Number of registrations published in the annual report (MB1 series) and currently on the National Cancer Registry database, England, 1971–2010
Image .csv .xls10. Cancer registrations: interpretation and data quality
It is estimated that the 2011 cancer registrations reported in this publication are around 98% complete (see Background Note 5). It is expected that the remaining 2% of cancer registrations for 2011 will be received over the next few years.
Some cancer registries are not always able to collect complete information about benign, uncertain and unknown neoplasms and therefore these registration rates are almost certainly underestimates of the true incidence. This should be noted when interpreting regional differences.
A specific example of variation in completeness of registration by cancer registry applies to non-melanoma skin cancers (nmsc, ICD-10 C44). In 2012, the South West Cancer Intelligence Service (SWCIS) reported that, due to national work priorities, there was a delay in completing registrations for nmsc for diagnosis year 2010. As a result, the number of registrations of nmsc for diagnosis year 2010 at the time of submission of cases to ONS was low. Registrations for 2010 nmsc were subsequently completed in mid-March 2012, after the cut-off date for publication of the 2010 Cancer Registrations Annual Reference Volume (number 41). The count of 2010 nmsc (ICD-10 C44) at completion was 20,050 registrations. This is reflected in this report for diagnosis year 2011 (see Table 8 (1.19 Mb Excel sheet)).
ONS has been advised by expert epidemiologists and members of the former Steering Committee on Cancer Registration that non-melanoma skin cancer (ICD-10 C44) and carcinoma in situ of skin (ICD-10 D04) are greatly under registered. Registration varies widely depending on a registry’s degree of access to out-patient and general practitioners records. This should be noted when interpreting these figures, particularly when considering changes over time.
During 2012, the English cancer registries embarked on a process to migrate to one national Encore cancer registration system. Use of this single system will ensure that all cancer registries record cancer incidence data using a consistent approach. Four of the registries completed migration in 2012, with the remaining four undergoing migration during 2013. Despite the advantages of a single national cancer registration system, the migration process created a considerable additional workload for registries. Some registries reported that this resulted in a lack of time for quality assurance work to be carried out as thoroughly as in previous years. It is possible, therefore, that some fluctuations in cancer registrations will be partly as a result of the change in cancer registration practice, and partly due to insufficient time for quality assurance work.
Due to the additional workload experienced by some of the cancer registries, the date for registrations for those patients diagnosed with cancer during 2011 to be received by ONS was delayed by two months: all registrations were submitted to ONS by the end of February 2013, instead of the end of December 2012 as would usually have been the case.
The agreed procedures to be followed by the cancer registries and ONS, when submitting and processing data, are set out in the ‘Registry/ONS Interface Document’ (Office of Population Censuses and Surveys, 1994). These are summarised by three levels of quality status, where levels one and two can be included in tables. It is a requirement that when a registry’s data for a particular year are complete, no more than 0.5% of records should have a quality recorded as Status 3, and over the past 12 years the proportion of records with serious errors has consistently been 0.1% or less. The quality status of all the records on the ONS cancer registration database from 1971 up to 2011 is shown in Table 10 (1.19 Mb Excel sheet) of the reference tables associated with this publication. Further details about the cancer registration data can be found in the Cancer Registration Statistics Quality and Methodology Information paper (244.2 Kb Pdf) and in previous Annual Reference Volumes.
Further comments on data quality and interpretation for this data can be found in the Background Notes and in the metadata with the tables accompanying this report (1.19 Mb Excel sheet).
Back to table of contents11. Users and uses of cancer statistics
Key users of cancer survival estimates include the Department of Health, academics and researchers, cancer charities, cancer registries, other government organisations, researchers within ONS, the media, and the general public. The Department of Health uses cancer statistics to brief parliamentary ministers, and as part of the evidence base to inform cancer policy and programmes, for example in drives to improve survival rates. Academics and researchers use the figures to inform their own research. Similarly cancer registries and other government organisations use the figures to carry out individual and collaborative projects to apply subject knowledge to practice. Charities use the data so they can provide reliable and accessible information about cancer to a wide range of groups, including patients and health professionals via health awareness campaigns and cancer information leaflets/web pages. Within the Office for National Statistics (ONS), cancer registration data are used to produce National Statistics on cancer incidence and survival. These data are also used to answer parliamentary questions and provide bespoke tables for customers.
Back to table of contents12. Policy context
In ‘Improving Outcomes: A Strategy for Cancer’ (January 2011), the Department of Health stated that although improvements have been made in the quality of cancer services in England, significant gaps remain in mortality and survival rates compared with the European average.
The Outcomes Strategy sets out how the Department of Health aims to improve outcomes for all cancer patients and improve cancer survival rates, with the aim of saving an additional 5,000 lives every year by 2014/15.
Outcomes strategies set out how the NHS, public health and social care services will contribute to the ambitions for progress agreed with the Secretary of State in each of the high-level outcomes frameworks. The indicators set for the NHS Outcomes Framework 2013 to 2014 – focus on measuring health outcomes include one- and five-year cancer survival indicators for all cancers combined, and for colorectal, breast and lung cancers combined.
Back to table of contents